BSL‑3: Design, Construction & Safe Operation of High‑Containment Laboratories
A Complete Engineering & Biosafety Guide for Designing and Operating Biosafety Level‑3 Facilities
Biosafety Level 3 (BSL‑3) laboratories are critical infrastructures for studying airborne Risk Group 3 pathogens and producing vaccines, diagnostics, and therapeutics that protect global public health. This book provides a complete, multidisciplinary roadmap for designing, constructing, validating, and safely operating BSL‑3 facilities in alignment with international standards such as WHO, CDC BMBL, ISO 14644, FDA, and EMA guidelines.
As stated in the book:
“The objective of this book is to provide a comprehensive and practical reference for the design, construction, and safe operation of Biosafety Level 3 (BSL‑3) laboratories…”
This work bridges architecture, HVAC engineering, biosafety, GMP compliance, and emerging technologies—offering a unified, practical reference for professionals across the entire facility lifecycle.
What Problem Does This Book Solve?
Designing and operating a BSL‑3 facility is one of the most complex engineering and biosafety challenges in the world. This book addresses the critical gaps that often lead to delays, safety failures, or regulatory non‑compliance:
-
Lack of integrated guidance combining architecture + HVAC + biosafety + GMP
-
Misalignment between biosafety requirements and pharmaceutical manufacturing needs
-
Insufficient understanding of airflow, pressure control, and filtration systems
-
Fragmented documentation and validation processes
-
Limited resources on emerging technologies (BIM, IoT, Digital Twin, robotics)
-
High operational costs due to inefficient HVAC and energy systems
This book provides clear, actionable, and standards‑based solutions to each of these challenges.
This book is designed for professionals involved in the planning, execution, and operation of high‑containment laboratories, including:
-
Mechanical and HVAC engineers
-
Architects and cleanroom designers
-
Biosafety officers and laboratory managers
-
GMP and quality assurance specialists
-
EPC/EPCM project teams
-
Researchers working with airborne pathogens
-
Regulatory and compliance professionals
Whether you are designing a new BSL‑3 facility or upgrading an existing one, this book serves as a trusted reference.
Why This Book Is Different
This book stands out because it:
-
Integrates biosafety, engineering, architecture, and GMP into a single coherent framework
-
Covers both research-focused and GMP‑compliant manufacturing BSL‑3 facilities
-
Provides detailed explanations of HVAC design, pressure regimes, HEPA/ULPA filtration, and validation
-
Includes risk management, emergency response, and waste handling protocols
-
Explores emerging technologies such as BIM, IoT, Digital Twin, robotics, and quantum AI
-
Addresses sustainability and low‑energy HVAC strategies for high‑containment labs
-
Offers practical tools including checklists, glossaries, and curated references
It is not just a theoretical text — it is a practical, standards‑driven guide for real‑world projects.
Chapter‑by‑Chapter Overview
Chapter 1 — Introduction to BSL‑3 Laboratories
-
Definition and purpose of BSL‑3 facilities
-
Comparison with BSL‑1, BSL‑2, and BSL‑4
-
Risk Group 3 pathogens (e.g., Mycobacterium tuberculosis, SARS‑CoV‑2)
-
Applications in research, vaccine development, and infectious disease control
Chapter 2 — Architectural Design Fundamentals
-
Airtight construction, seamless surfaces, chemical‑resistant materials
-
Clean / transition / dirty zoning
-
Personnel flow and access control
-
Anterooms, airlocks, and pressure cascades
Chapter 3 — HVAC Systems for BSL‑3
-
Negative pressure differentials (≥12.5 Pa)
-
HEPA/ULPA filtration and Bag‑In/Bag‑Out housings
-
Air change rates (6–12 ACH)
-
Redundancy, monitoring, and emergency power
-
Validation: DOP testing, airflow verification, ACH measurement
Chapter 4 — Biosafety Equipment
-
Class II and Class III biosafety cabinets
-
PPE and donning/doffing procedures
-
Emergency showers and eyewash stations
-
Robotics and automation for high‑risk tasks
Chapter 5 — GMP Compliance in BSL‑3 Facilities
-
Cleanroom classification (ISO 5–7)
-
Contamination control and documentation
-
DQ/IQ/OQ/PQ validation
-
LIMS, QMS, blockchain records, and 21 CFR Part 11 compliance
Chapter 6 — Safety Protocols and Risk Management
-
Biological risk assessment (hazard identification, FMEA, FTA)
-
Emergency response for spills, aerosol events, HVAC failures
-
Waste management and traceable disposal
-
AR/VR training and simulation‑based competency building
Chapter 7 — Emerging Technologies
-
BIM for design coordination and lifecycle management
-
Digital Twin for real‑time monitoring and predictive maintenance
-
IoT sensors and AI‑driven optimization
-
3D printing for custom sterilizable components
-
Quantum AI for advanced pathogen analysis
Chapter 8 — Sustainability in BSL‑3 Design
-
Low‑energy HVAC strategies
-
Heat recovery and EC fans
-
Renewable energy integration
-
LEED/BREEAM alignment
-
Reducing carbon footprint without compromising biosafety
Chapter 9 — The Future of BSL‑3 Laboratories
-
Pandemic preparedness
-
Gene therapy and advanced biomanufacturing
-
Modular and scalable BSL‑3 designs
-
Global harmonization of standards
-
Space biotechnology and extraterrestrial containment
Selected Key Quotes from the Book
“BSL‑3 laboratories must integrate rigorous biosafety, engineering, and quality systems to protect personnel, the public, and product integrity.”
“HVAC systems are the backbone of BSL‑3 containment, ensuring stable negative pressure, validated filtration, and controlled environmental conditions.”
This book is an essential reference for anyone involved in the design, construction, or operation of BSL‑3 facilities. It brings together engineering, biosafety, GMP, and emerging technologies into a single, authoritative guide.
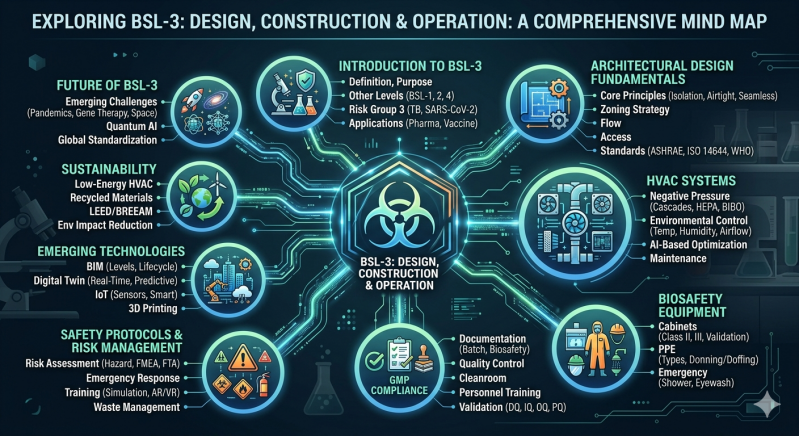
BSL‑3: Design, Construction & Operation
│
├── 1. Introduction to BSL‑3
│ ├── Definition & Purpose
│ ├── Comparison with Other Levels
│ │ ├── BSL‑1
│ │ ├── BSL‑2
│ │ └── BSL‑4
│ ├── Risk Group 3 Microorganisms
│ │ ├── Characteristics
│ │ ├── Examples (TB, SARS‑CoV‑2)
│ │ └── Handling Requirements
│ └── Applications
│ ├── Pharmaceuticals
│ ├── Vaccine Development
│ └── Infectious Disease Research
│
├── 2. Architectural Design Fundamentals
│ ├── Core Principles
│ │ ├── Isolation
│ │ ├── Airtight Construction
│ │ └── Seamless Surfaces
│ ├── Zoning Strategy
│ │ ├── Clean Zone
│ │ ├── Transition Zone
│ │ └── Dirty Zone
│ ├── Personnel Flow
│ ├── Access Control
│ └── Standards
│ ├── ASHRAE
│ ├── ISO 14644
│ └── WHO Guidelines
│
├── 3. HVAC Systems
│ ├── Negative Pressure Systems
│ │ ├── Pressure Cascades
│ │ ├── HEPA/ULPA Filtration
│ │ └── BIBO Systems
│ ├── Environmental Control
│ │ ├── Temperature
│ │ ├── Humidity
│ │ └── Airflow
│ ├── AI‑Based Optimization
│ │ ├── Predictive Control
│ │ └── Energy Efficiency
│ └── Maintenance & Validation
│ ├── DOP Testing
│ ├── ACH Verification
│ └── Monitoring Systems
│
├── 4. Biosafety Equipment
│ ├── Biosafety Cabinets
│ │ ├── Class II
│ │ ├── Class III
│ │ └── Validation
│ ├── PPE
│ │ ├── Types
│ │ └── Donning/Doffing
│ └── Emergency Systems
│ ├── Shower
│ └── Eyewash
│
├── 5. GMP Compliance
│ ├── Documentation
│ │ ├── Batch Records
│ │ ├── Biosafety Records
│ │ └── QC Records
│ ├── Quality Control
│ ├── Cleanroom Classification
│ ├── Personnel Training
│ └── Validation
│ ├── DQ
│ ├── IQ
│ ├── OQ
│ └── PQ
│
├── 6. Safety Protocols & Risk Management
│ ├── Biological Risk Assessment
│ │ ├── Hazard Identification
│ │ ├── FMEA / FTA
│ │ └── Documentation
│ ├── Emergency Response
│ │ ├── Spills
│ │ └── HVAC Failures
│ ├── Personnel Training
│ │ ├── Simulation
│ │ └── AR/VR
│ └── Waste Management
│ ├── Waste Types
│ └── Disposal Procedures
│
├── 7. Emerging Technologies
│ ├── BIM
│ │ ├── Levels
│ │ ├── Coordination
│ │ └── Lifecycle Management
│ ├── Digital Twin
│ │ ├── Real‑Time Monitoring
│ │ └── Predictive Maintenance
│ ├── IoT
│ │ ├── Sensors
│ │ └── Smart Monitoring
│ └── 3D Printing
│ ├── Custom Components
│ └── Sterilizable Materials
│
├── 8. Sustainability
│ ├── Low‑Energy HVAC
│ ├── Recycled Materials
│ ├── LEED/BREEAM
│ └── Environmental Impact Reduction
│
└── 9. Future of BSL‑3
├── Emerging Challenges
│ ├── Pandemics
│ ├── Gene Therapy
│ └── Space Research
├── Quantum AI
└── Global Standardization
Why "Containment" Is Paramount
A Specialized Approach to Biosecurity and Public Health Protection
Introduction
For decades, the primary objective of biosafety laboratory design has been summarized in one word: containment. Standards such as CDC, WHO, and NIH have shaped the modern high-containment environment by defining acceptable levels for facility design, operational practices, and safety equipment.
Yet a fundamental question remains unanswered:
If a BSL-3 laboratory meets every conventional containment standard, why do incidents, near-misses, or public concerns still arise?
This question is the starting point of the BSL-3 framework.
Foundational Questions BSL-3 Puts on the Table
Does biosafety extend only to primary barriers and HEPA filtration?
Or does it also encompass human factors, organizational culture, emergency preparedness, and the evolving threat landscape?
Is compliance a sufficient indicator of biosecurity?
Or should we treat proactive risk management as a primary operational target?
Do current standards capture all relevant variables for emerging pathogens?
Or is a significant portion of potential risks still unaddressed or evolving?
BSL-3 answers these questions with scientific clarity, engineering precision, and practical protocols.
The Limitations of the Traditional "Compliance" Paradigm
Compliance is a baseline, not a guarantee of safety
A facility may meet every regulatory checklist while the environment simultaneously presents risks from:
-
undetected breaches in negative pressure
-
human error in PPE donning/doffing
-
unforeseen equipment malfunction
-
evolving characteristics of novel pathogens
These factors are not always consciously perceived,
but they can influence worker safety, public health, research integrity, and institutional reputation.
Conventional standards measure only a fraction of what truly matters
HEPA filtration, airflow rates, and decontamination protocols are important,
but they represent only a small portion of the variables that affect biosecurity in a BSL-3 setting.
Current standards do not fully address:
-
human factors and cognitive load
-
dynamic risk assessment for novel agents
-
the psychology of compliance and complacency
-
sophisticated cyber-biosecurity threats
-
complex interdependencies in mechanical systems
-
long-term sustainability of containment measures
-
global health security implications
BSL-3 integrates these missing variables into a unified operational and engineering framework.
A BSL-3 facility is a complex socio-technical system
Biocontainment relies not only on physical barriers,
but also on interconnected human actions and technological safeguards.
Procedures, training, and culture are not merely supplementary;
they are critical inputs that influence:
-
adherence to protocols
-
rapid incident response
-
effectiveness of engineering controls
-
overall security posture
-
public trust
This is the foundation of BSL-3.
Core Biocontainment Principles in BSL-3
Key equipment for primary containment includes:
-
Class II or III Biosafety Cabinets (BSCs)
-
Centrifuge safety cups/rotors
-
Sealed containers for transport and storage
-
Sharps containers & waste autoclaves
-
Personal Protective Equipment (PPE)
BSL-3 provides detailed methods for:
-
selection and validation of BSCs
-
proper use and maintenance of PPE
-
risk assessment for specific procedures
-
decontamination protocols for equipment
-
minimizing aerosol generation
In BSL-3, the facility itself is a critical barrier, not just a building.
The book explains:
-
how directional airflow and negative pressure prevent pathogen escape
-
how HEPA filtration of exhaust air ensures environmental protection
-
how robust, sealable surfaces facilitate decontamination
-
why dedicated HVAC systems are essential for safety
-
how to engineer facilities for operational redundancy and resilience
Human behavior is a critical factor in containment.
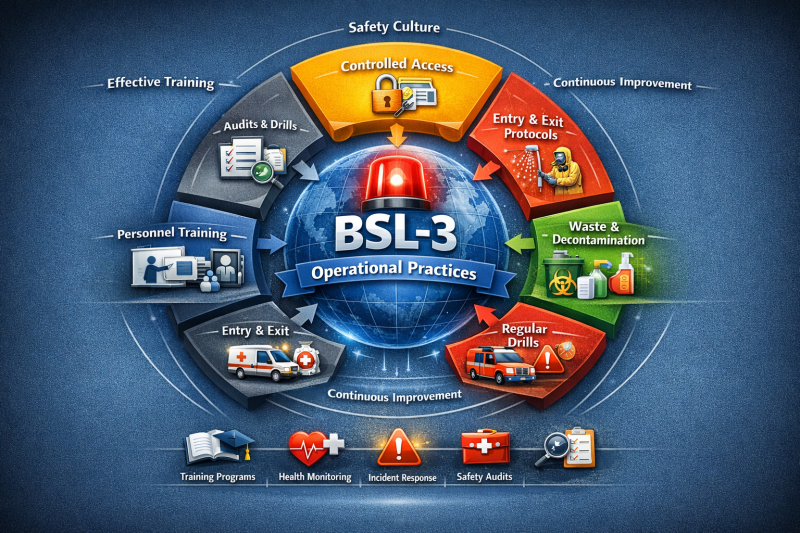
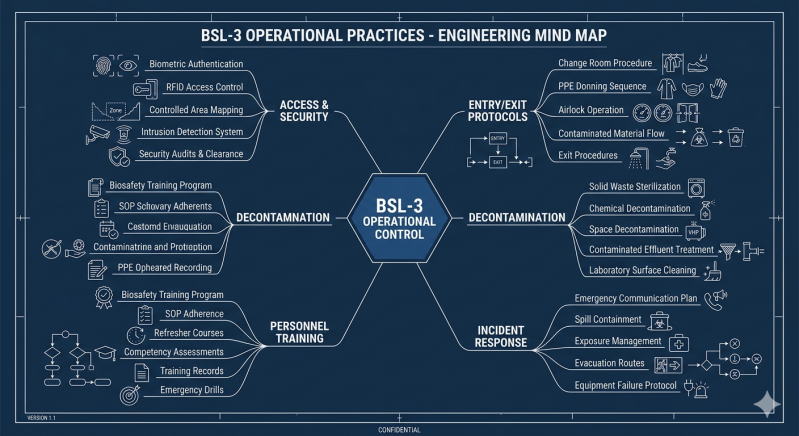
Operational practices in BSL-3 include:
-
controlled access & security systems
-
strict entry/exit procedures (e.g., showers, change rooms)
-
defined waste management & decontamination protocols
-
incident response & emergency plans
-
comprehensive personnel training & health surveillance
BSL-3 provides methods for:
-
developing effective training programs
-
fostering a strong safety culture
-
conducting regular drills and exercises
-
auditing and continuous improvement of practices
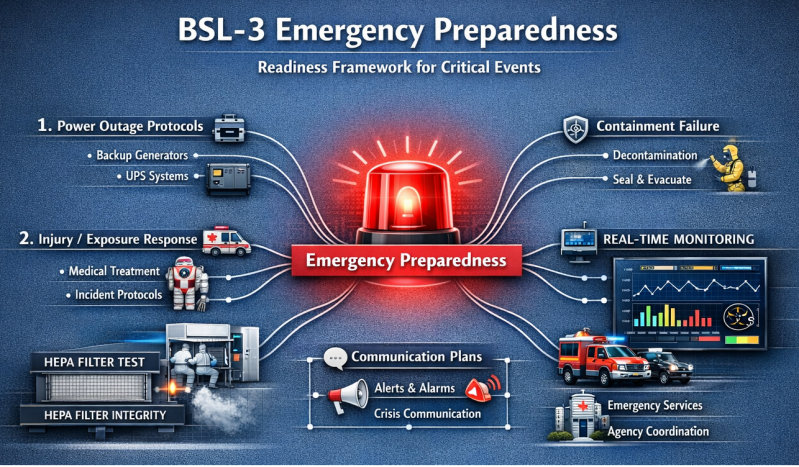
Readiness for unforeseen events is paramount in BSL-3.
The framework addresses:
-
power outage protocols & backup systems
-
containment failure scenarios
-
personnel injury or exposure response
-
external emergency services coordination
-
communication plans during incidents
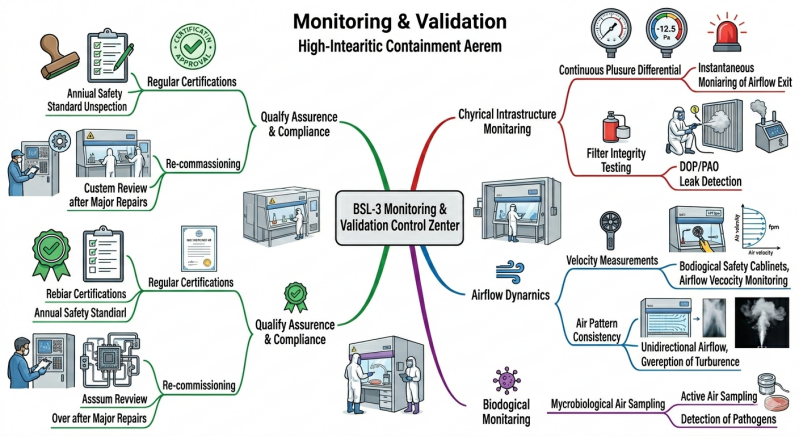
BSL-3 extends biosafety assurance into real‑time adaptive systems using:
-
continuous pressure differential monitoring
-
HEPA filter integrity testing (e.g., DOP/PAO)
-
airflow velocity measurements
-
microbiological air sampling
-
regular certification and re-commissioning
This transforms a BSL-3 lab into a continuously validated, high-integrity containment system.
Practical and Economic Outcomes
Enhanced Safety & Biosecurity
Optimized design, rigorous protocols, and continuous monitoring
minimize risks of pathogen release and exposure.
Reduced Incident Rates & Downtime
Proactive risk management and robust systems lead to
fewer accidents and operational interruptions.
Improved Public & Institutional Trust
Demonstrable commitment to safety and transparency
strengthens confidence in research activities.
Return on Investment
Institutions benefit from:
-
protection of personnel and community
-
preservation of valuable research materials
-
avoidance of costly remediation and legal liabilities
-
maintenance of research continuity and funding eligibility
BSL-3 is not simply another book on biosafety standards.
It is a comprehensive framework that redefines high-containment laboratories as integrated systems for pathogen control.
In this framework:
-
compliance is necessary but insufficient
-
proactive risk management becomes a measurable operational target
-
facility design, human factors, and emergency preparedness are equally critical
-
laboratories can actively contribute to global health security
For biosafety professionals and facility managers seeking a deeper understanding of next-generation high-containment design and operations,
BSL-3 provides the scientific foundation, engineering tools, and practical protocols needed to create environments that truly protect against biological threats.








Create Your Own Website With Webador